09 Nov, 2024 Design of Phase 1 Study of ALN-HTT02 in Adult Patients with Huntington’s Disease Presented at the Annual Meeting of the Huntington Study Group (HSG)
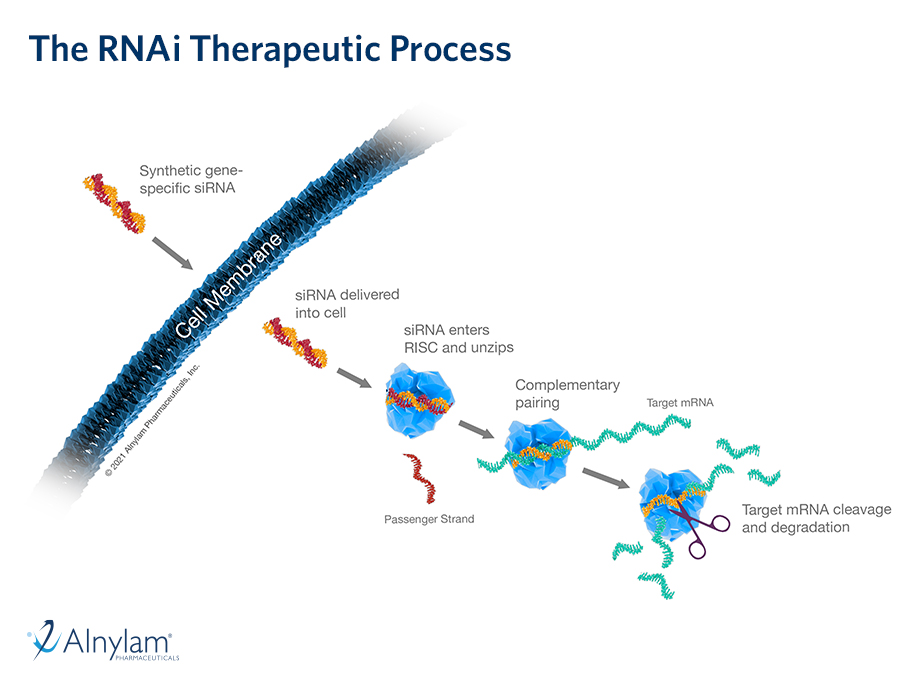
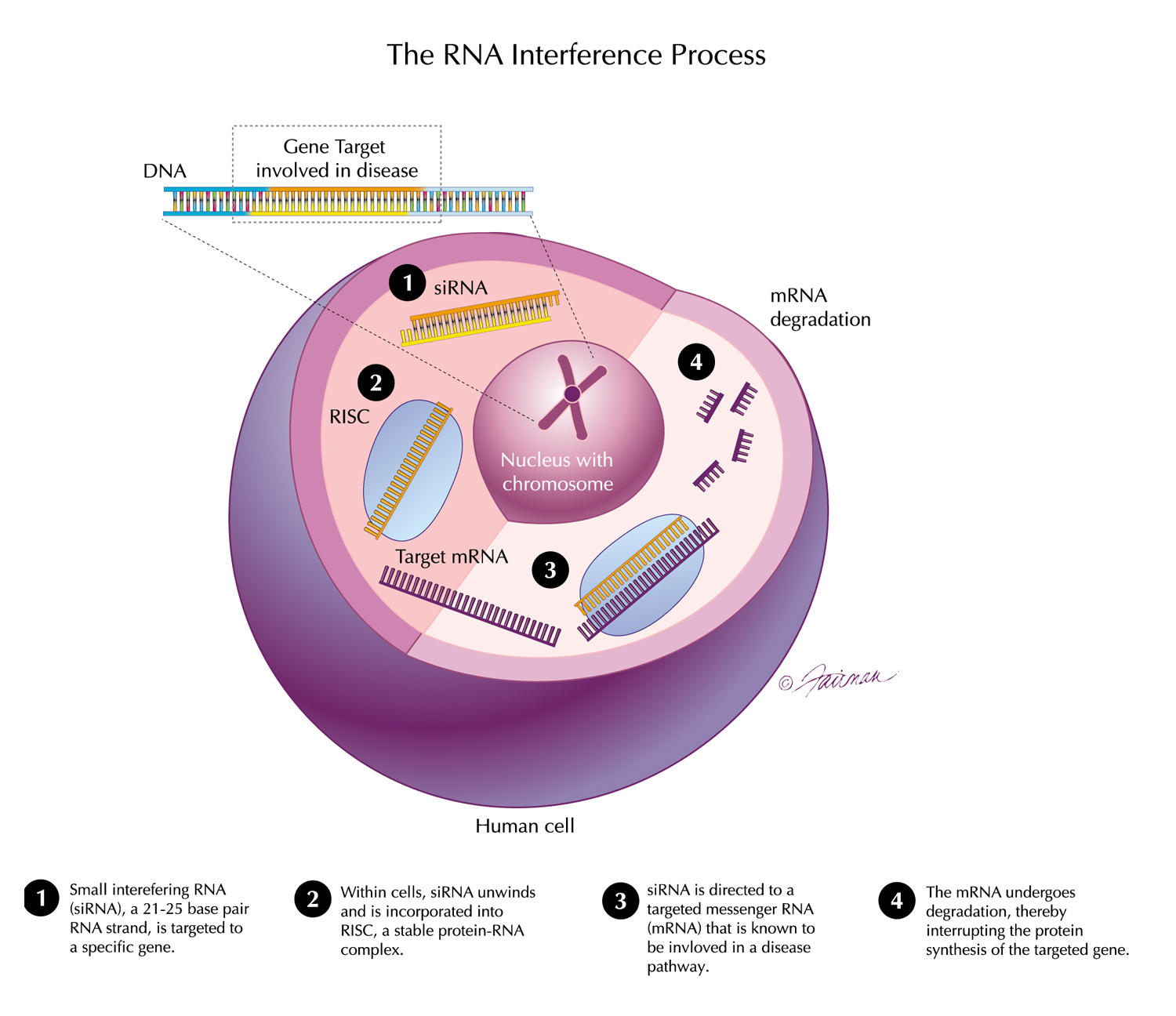
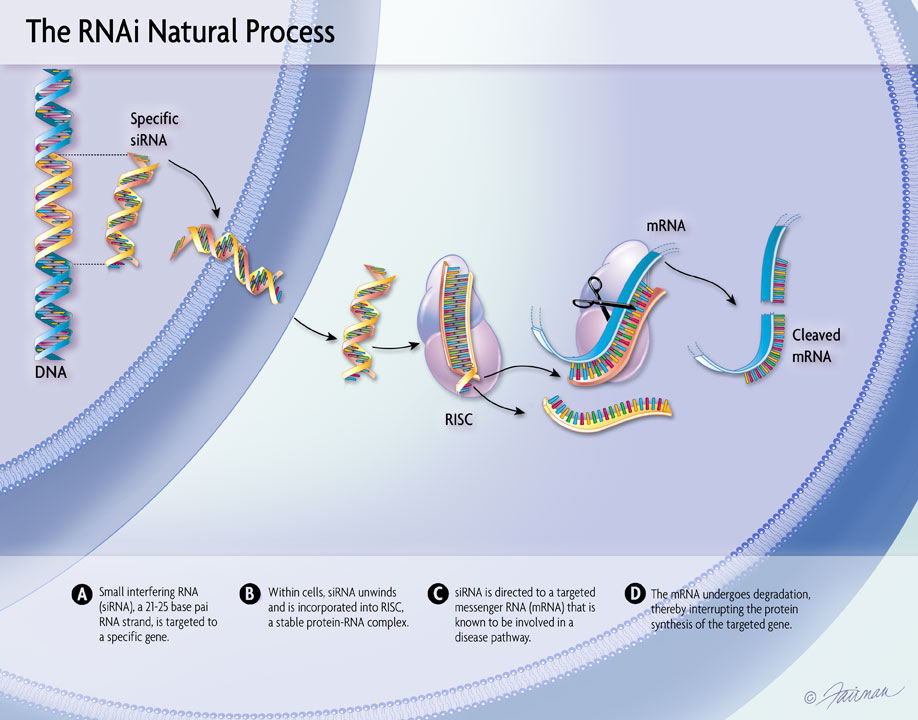
The design of a Phase 1 study of ALN-HTT02—an investigational RNAi therapeutic targeting huntingtin (HTT) for Huntington’s disease—was presented at the 31st Annual Meeting of the Huntington Study Group (HSG) in Cincinnati, Ohio.
Sloan, et al. “ALN-HTT02, a Novel C16-siRNA Conjugate for HTT-lowering in the CNS”