15 Jun, 2025 UK Biobank Analyses and Preclinical Data on Hepatic Plasminogen Lowering Presented at the 2025 European Hematology Association (EHA) Congress
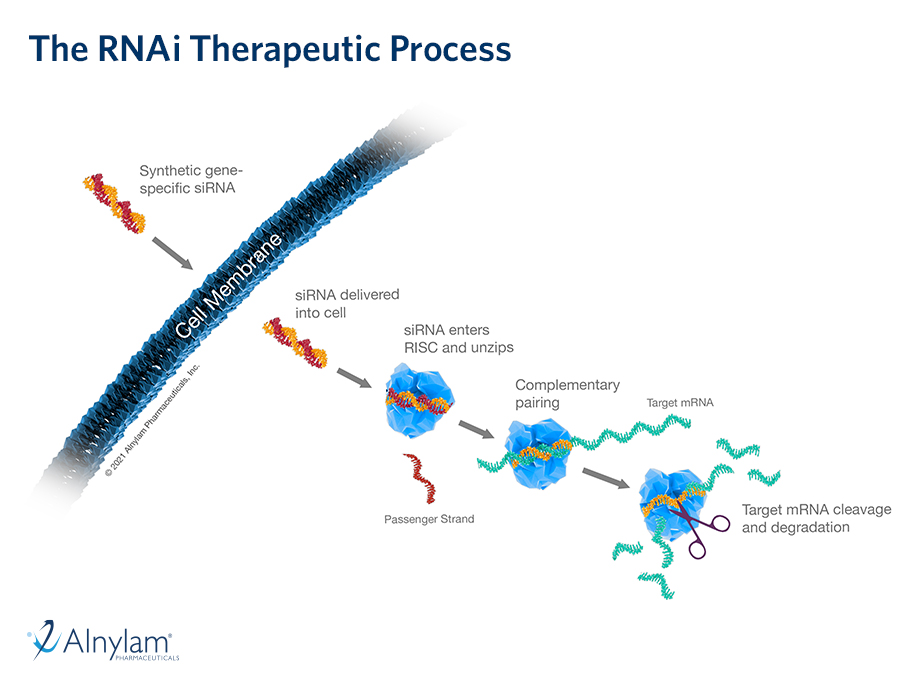
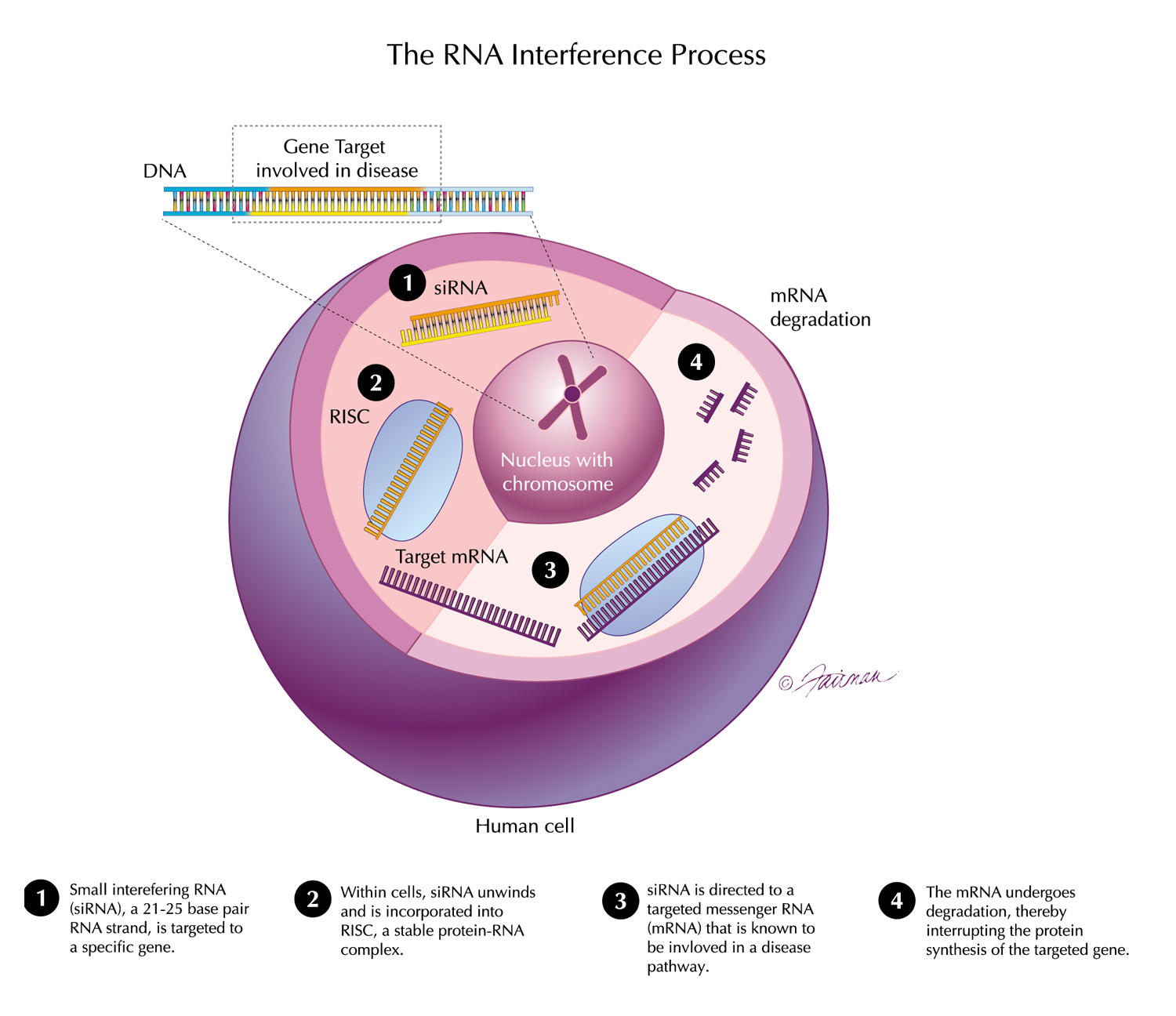
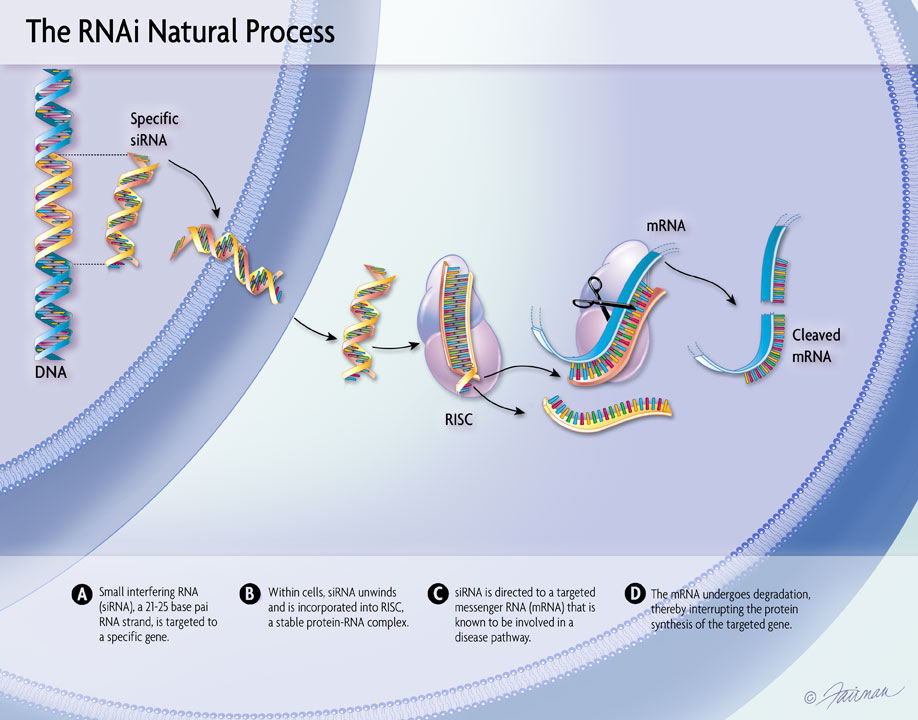
Human genetics and proteomics data from UK Biobank as well as data from preclinical models relevant to ALN-6400—an investigational RNAi therapeutic targeting plasminogen for bleeding disorders—were presented at the 2025 European Hematology Association (EHA) Congress, June 12-15, Milan, Italy.