20 Apr, 2026 TRITON-PN Phase 3 Study Design and Rationale Presented at the 2026 Annual American Academy of Neurology (AAN) Meeting
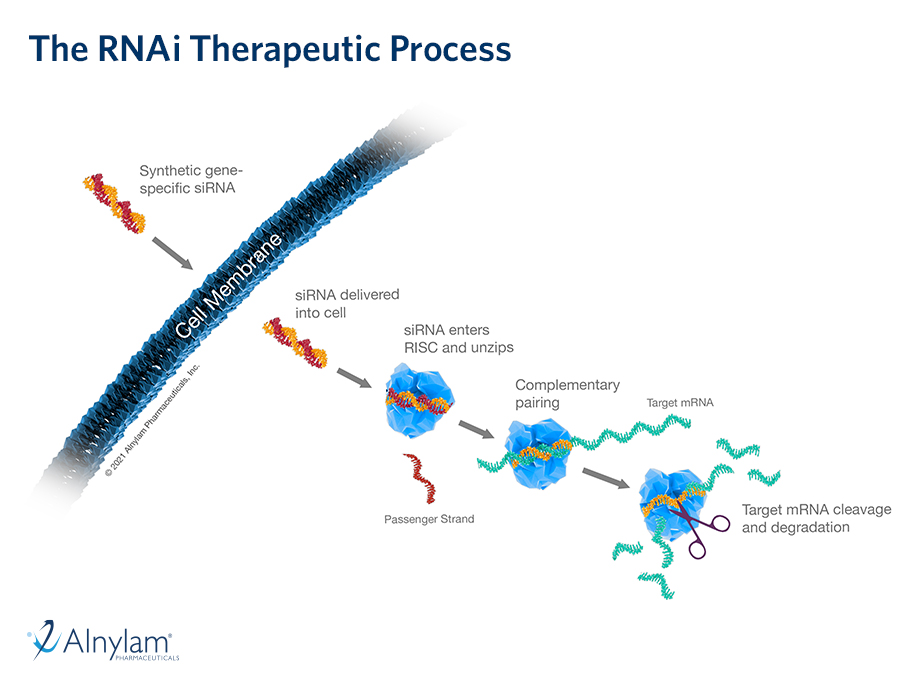
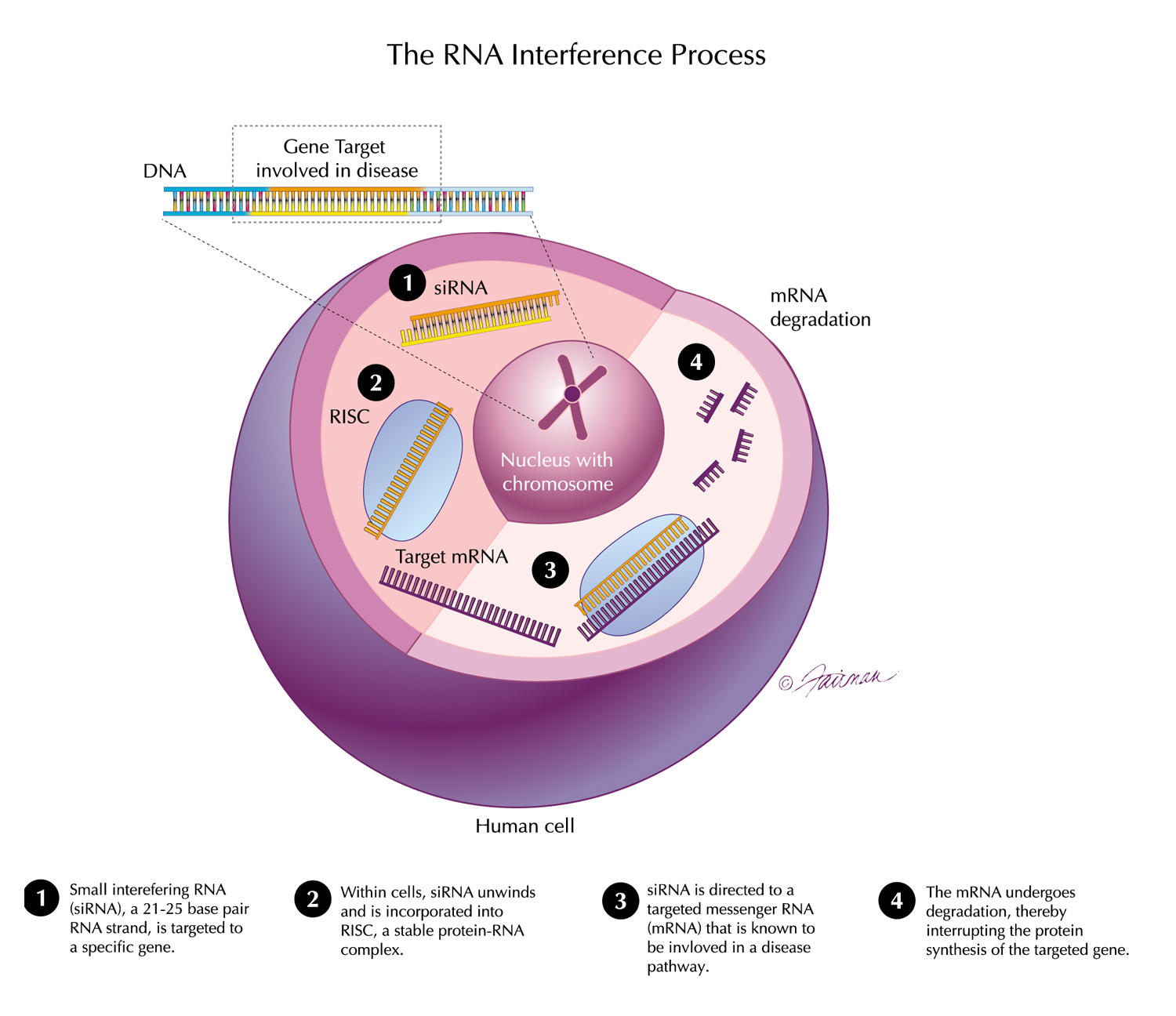
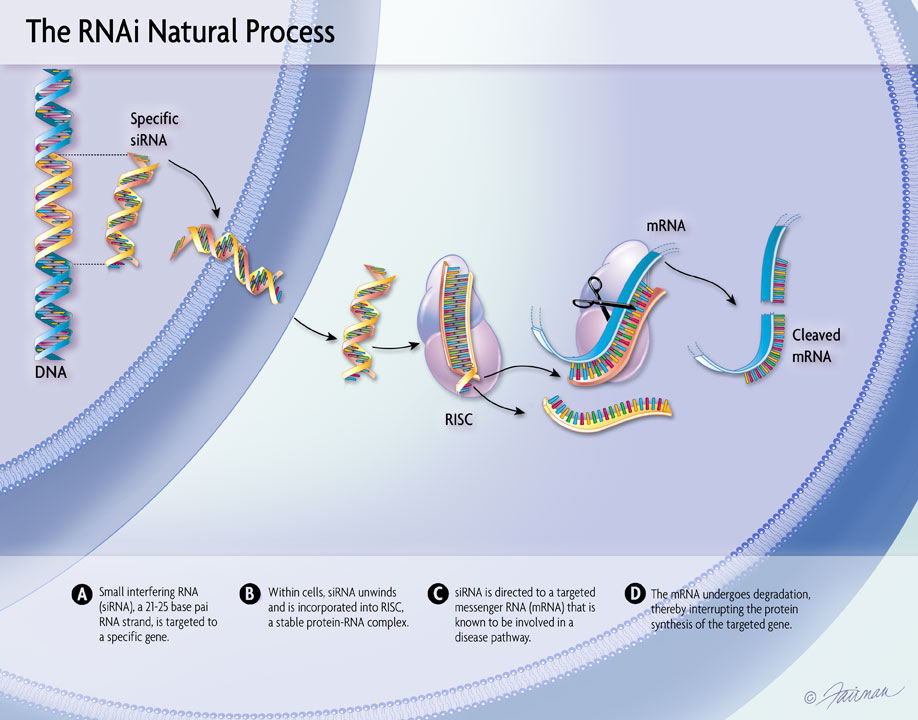
The design and rationale for the TRITON-PN Phase 3 study evaluating our third-generation investigational transthyretin (TTR) silencer, nucresiran, in patients with hereditary transthyretin amyloidosis with polyneuropathy (hATTR-PN) was presented at AAN 2026.