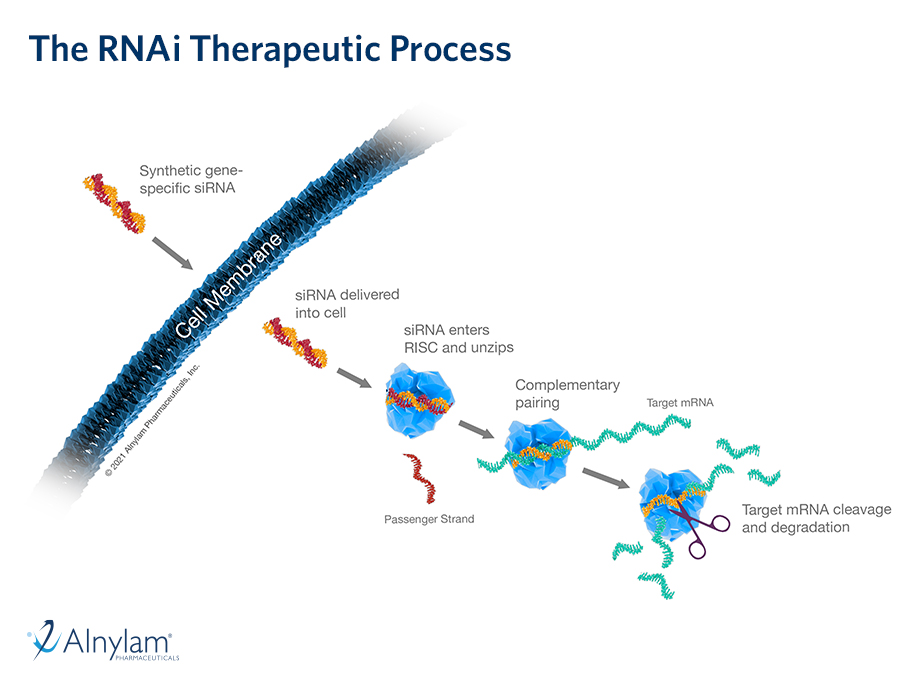
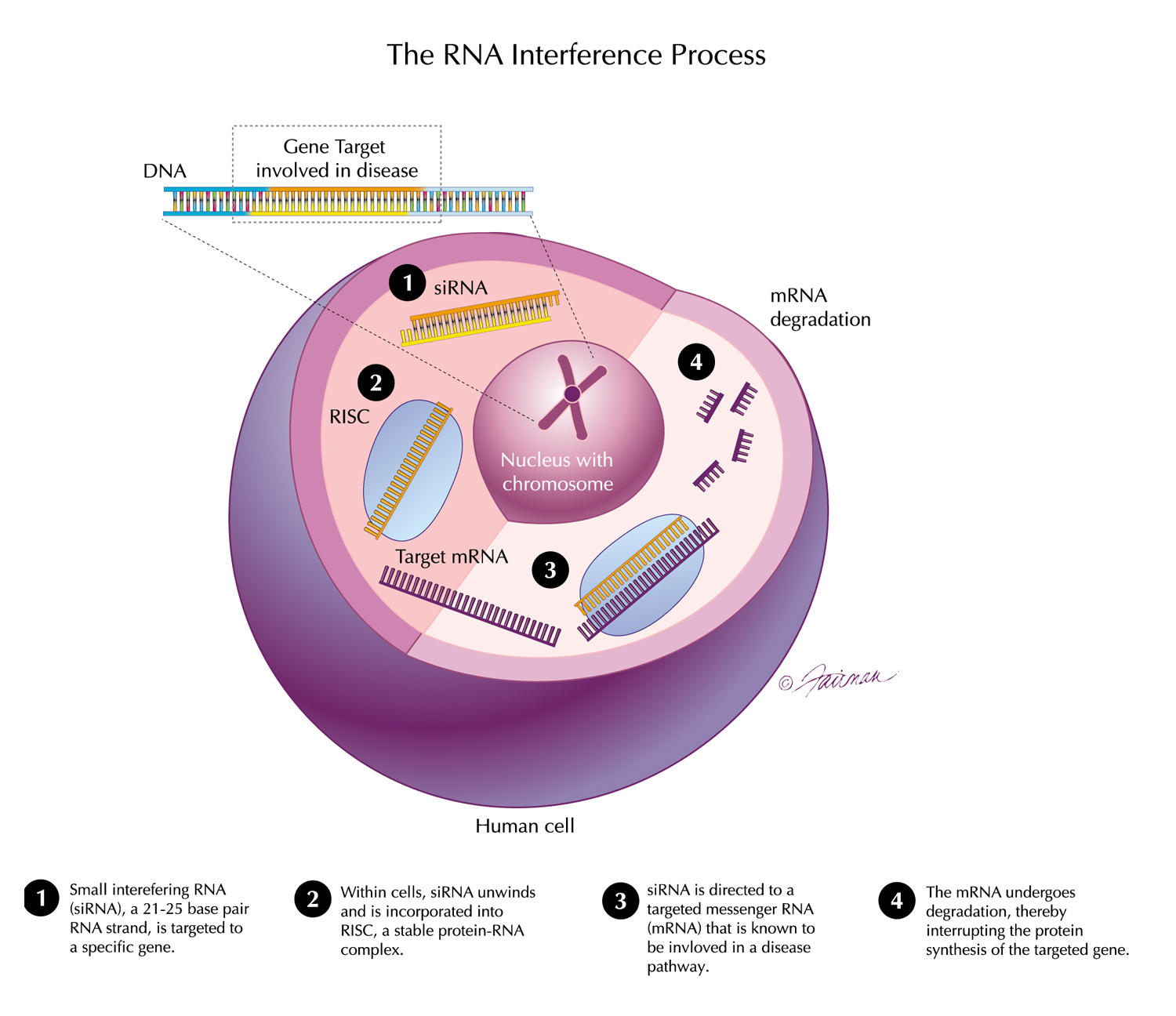
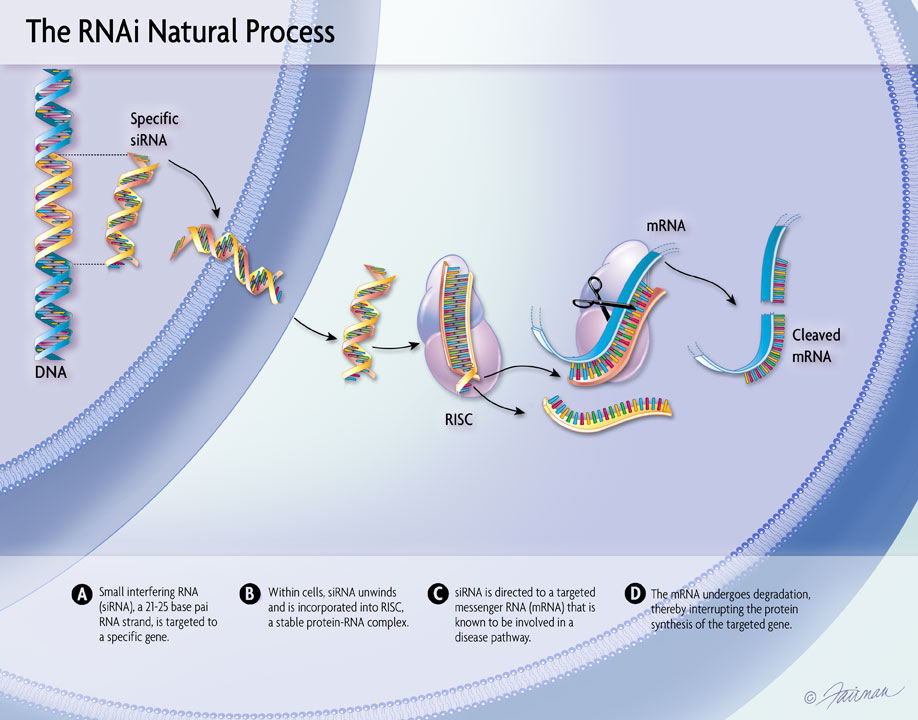
29 Mar, 2019 Pre-Clinical Data on Delivery of RNAi Therapeutics to the CNS and Eye
We presented new pre-clinical data on our efforts in CNS and ocular delivery of novel siRNA conjugates in rats and non-human primates at the sixth annual Cold Spring Harbor Conference on RNA and Oligonucleotide Therapeutics, held March 27 to 30 in Cold Spring Harbor, NY.