Posted at 07:36h
in
Capella
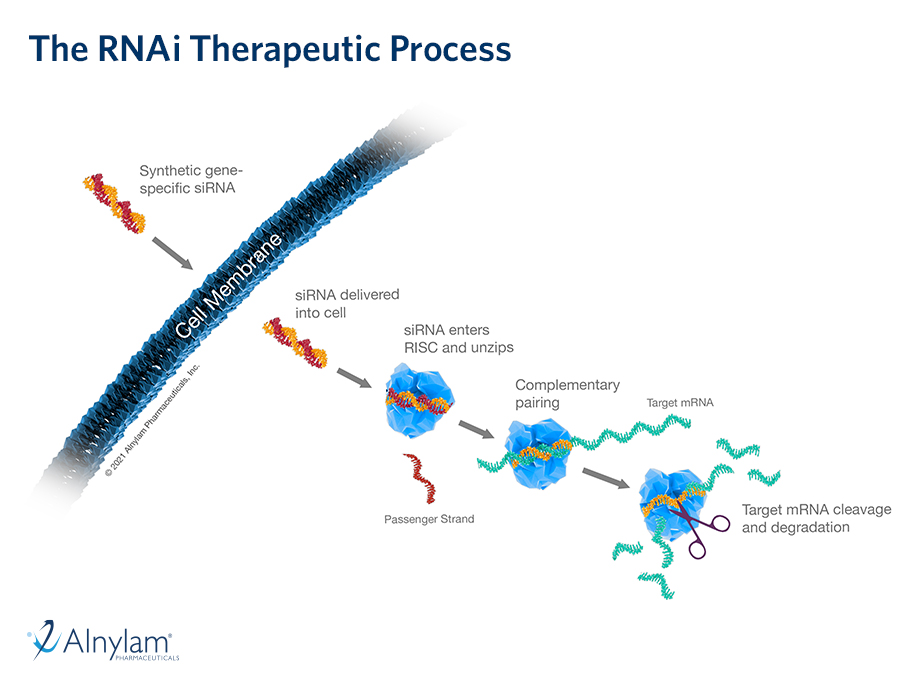
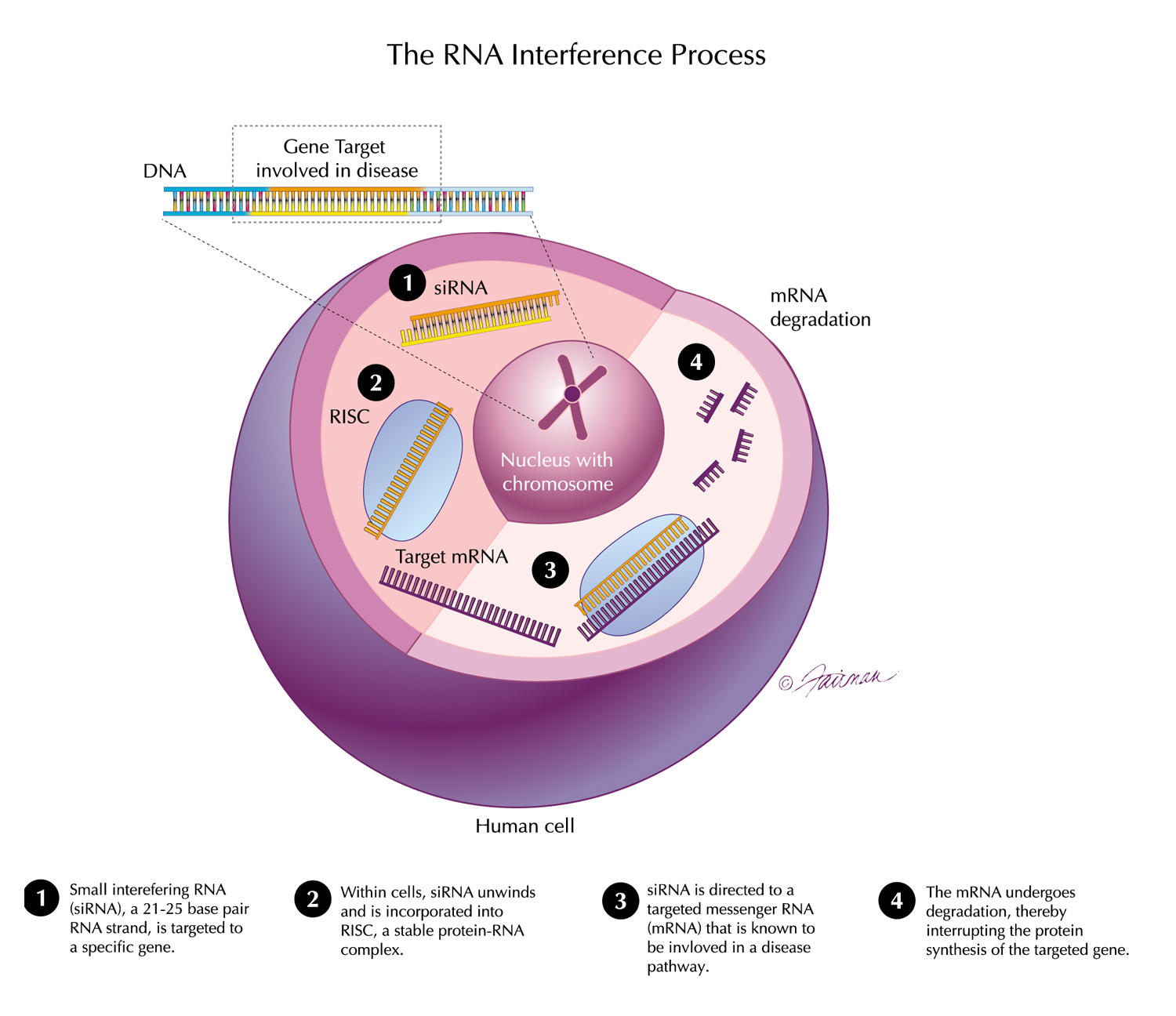
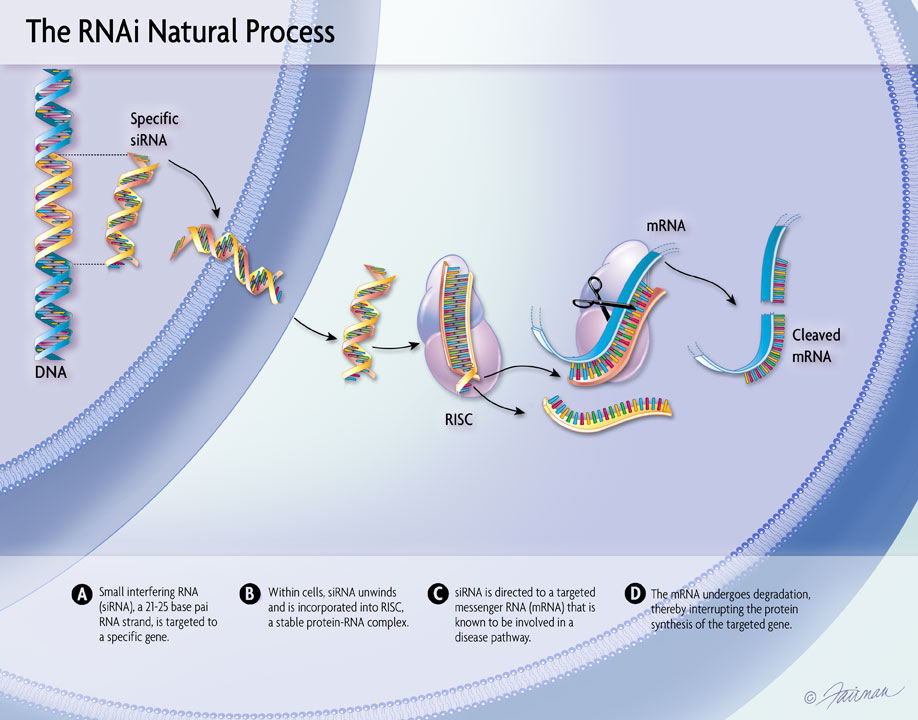
Results from pooled, post-hoc analyses from ongoing clinical trials of OXLUMO® (lumasiran), an RNAi therapeutic targeting hydroxyacid oxidase 1 – the gene encoding glycolate oxidase – for the treatment of primary hyperoxaluria type 1 (PH1), were presented at the 58th European Renal Association – European Dialysis and Transplant Association (ERA-EDTA) virtual congress, held June 5-8, 2021. The analyses found that treatment with lumasiran led to substantial and clinically meaningful reductions in urinary and plasma oxalate that were similar in pediatric and adult patients with PH1 in the ILLUMINATE-A and ILLUMINATE-B Phase 3 trials, with lumasiran demonstrating an acceptable safety profile in both patient populations. In additional pooled, post-hoc analyses, PH1 patients 12 months and older with a baseline estimated glomerular filtration rate (eGFR) of ≥30 mL/min/1.73m2, treated with lumasiran through Month 12, demonstrated stable eGFR levels, irrespective of baseline kidney function – an encouraging observation, given the progressive decline in kidney function characteristic of PH1.
Shasha-Lavsky, et al. “Lumasiran Demonstrated Comparable Oxalate Reduction and Safety in Children and Adults with Primary Hyperoxaluria Type 1”
Hayes, et al. “Stable eGFR in Patients With Primary Hyperoxaluria Type 1 Treated With Lumasiran, Regardless of Kidney Function at Start of Treatment”