30 Mar, 2026 New Analyses from Amyloidosis and Hypertension Programs Presented at The American College of Cardiology (ACC) Scientific Sessions 2026
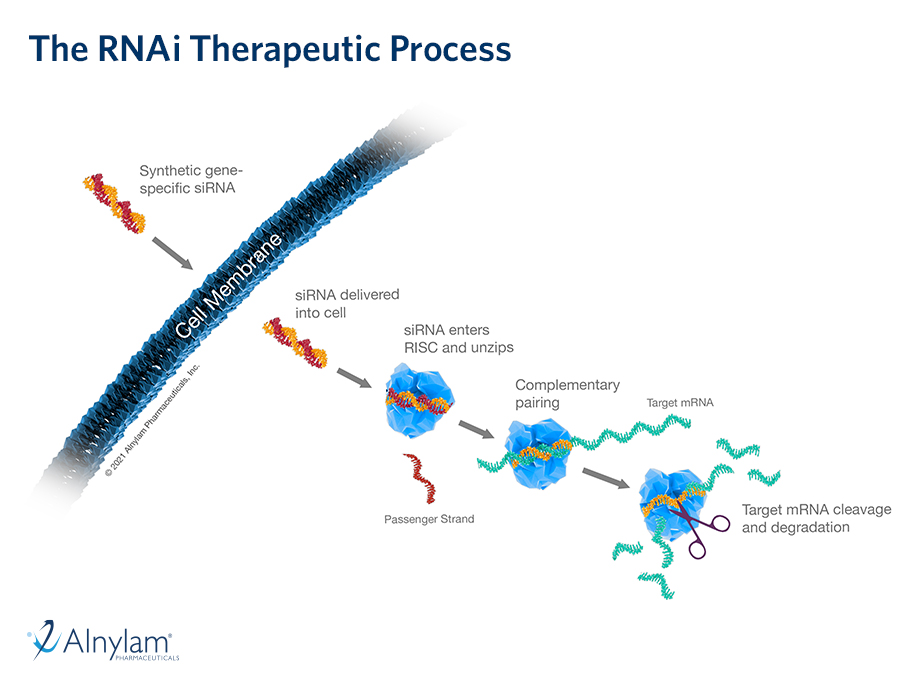
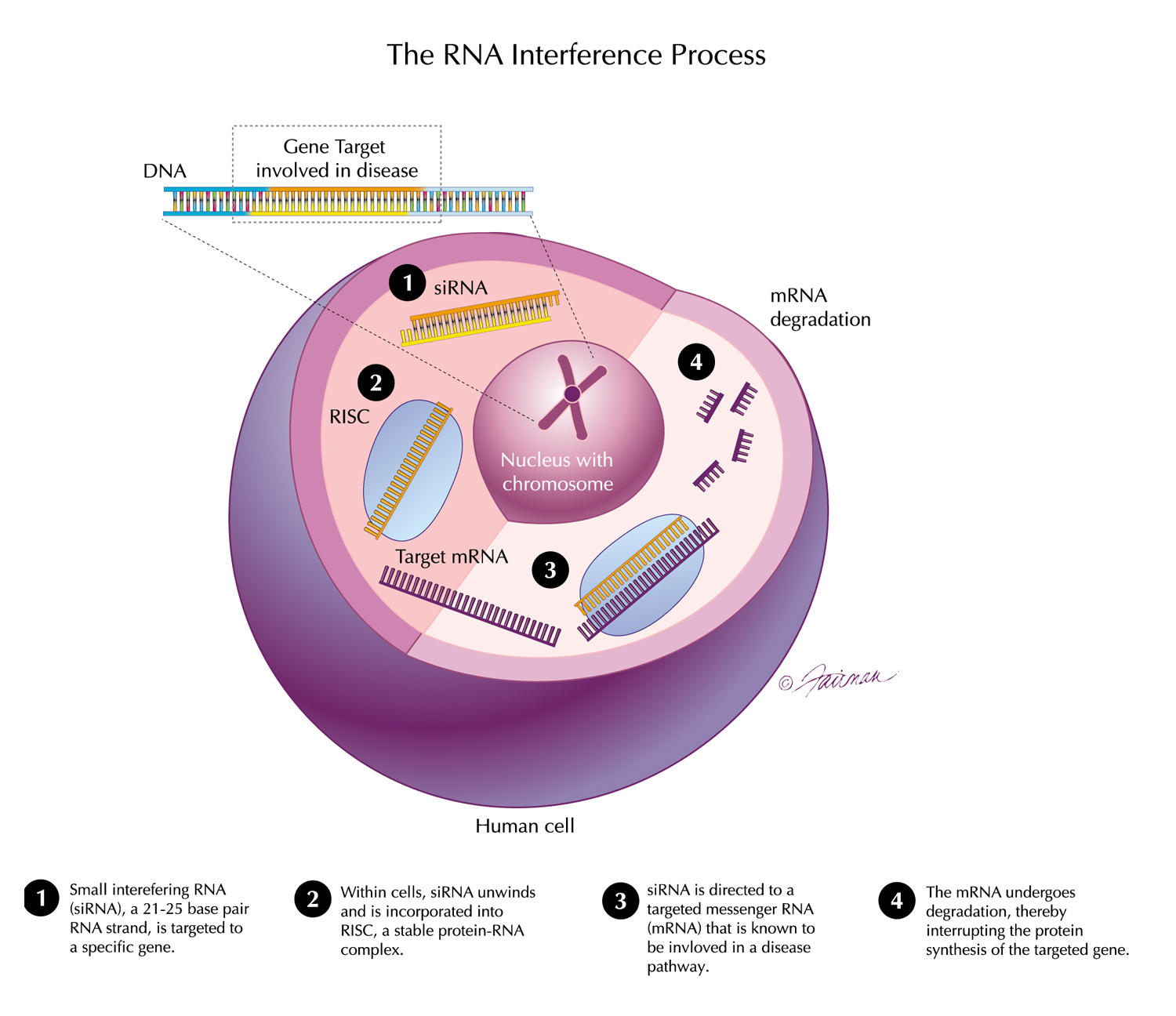
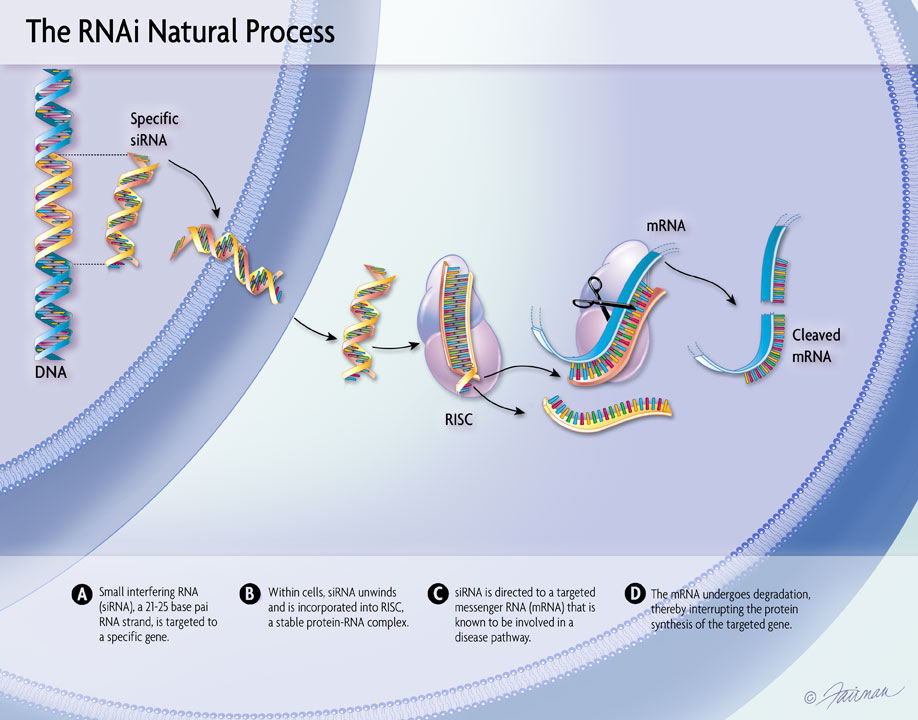
Analyses presented at ACC reinforce the totality of data for vutrisiran in patients with ATTR-CM including the impact on cardiovascular outcomes across a range of patient subgroups, including those with most advanced disease and diastolic dysfunction and on health-related quality of life. Real-world evidence also shows high treatment adherence with quarterly dosing.
A pooled safety analysis from the Phase 2 KARDIA studies demonstrate zilebesiran’s acceptable safety profile across a broad hypertension population, supporting continued investigation in the Phase 3 ZENITH cardiovascular outcomes trial.