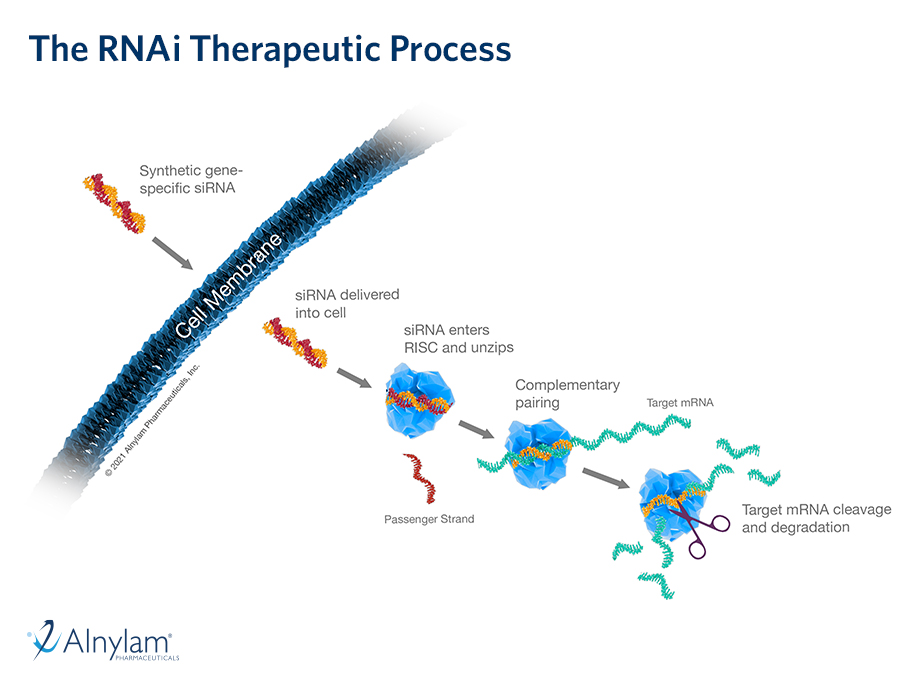
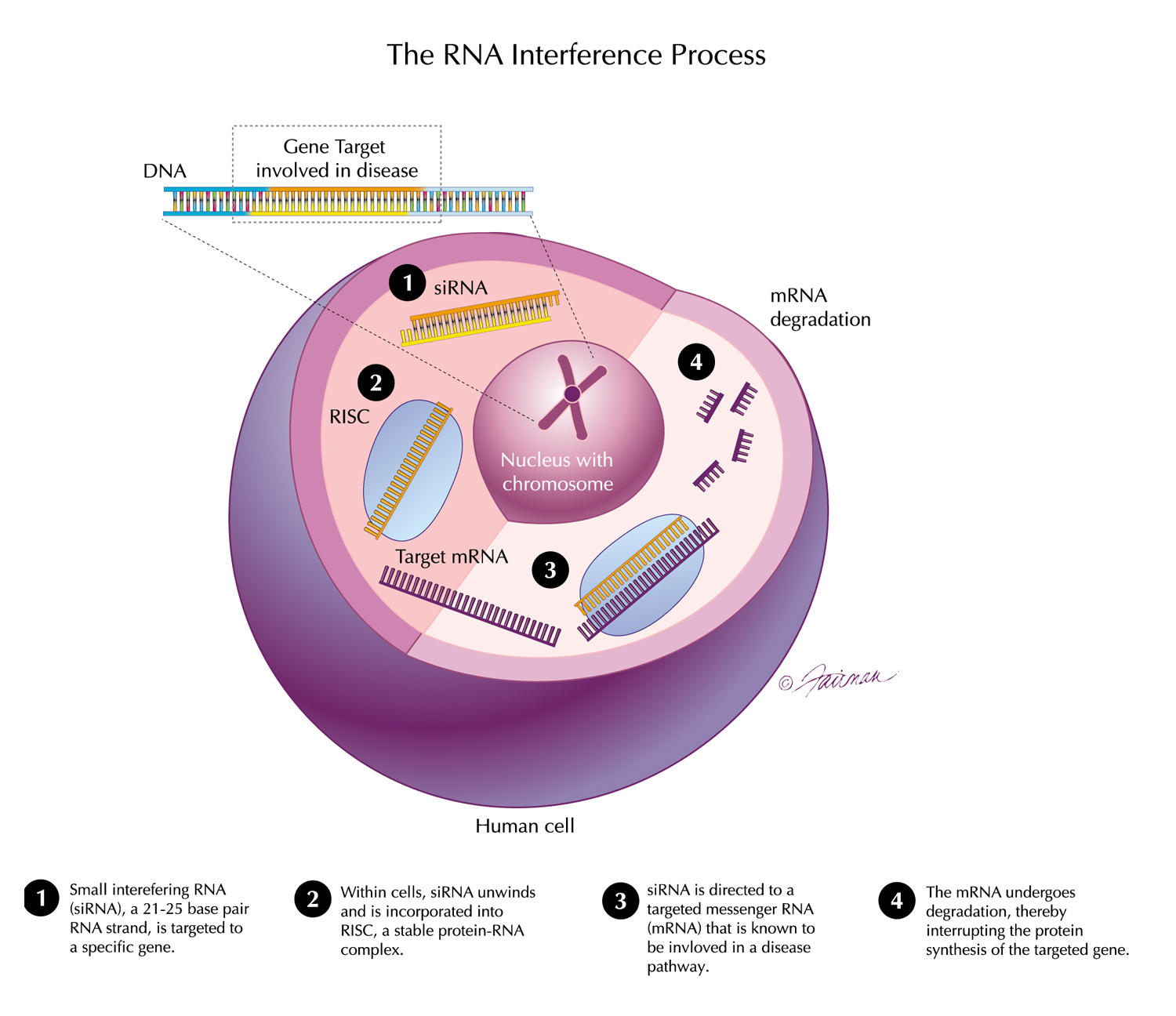
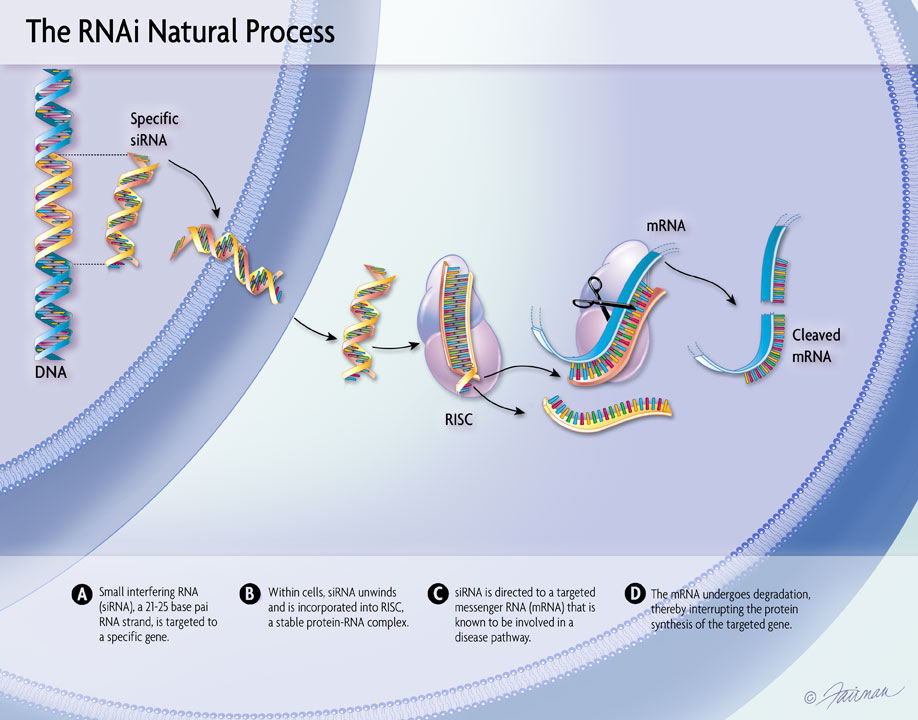
04 Oct, 2021 New Long-Term Results for GIVLAARI® (givosiran) Presented at UEG Week Virtual 2021
Positive findings from a 24-month interim analysis of the ongoing open-label extension (OLE) period of the ENVISION Phase 3 study of GIVLAARI® (givosiran) in patients with acute hepatic porphyria (AHP) were presented during the United European Gastroenterology (UEG) Week Virtual 2021 (Oct. 3-5). The data further confirm that long-term dosing with GIVLAARI provides sustained and continuous benefit to patients with AHP while maintaining an acceptable safety profile. Findings on the burden of AHP from the ENVISION Phase 3 study and from Part B of the EXPLORE study were also presented.