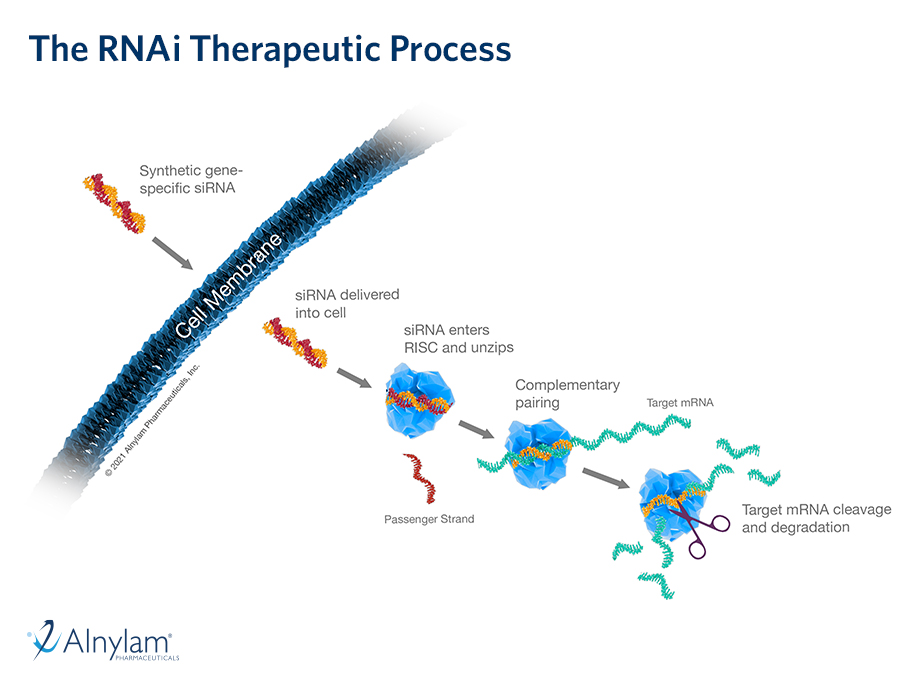
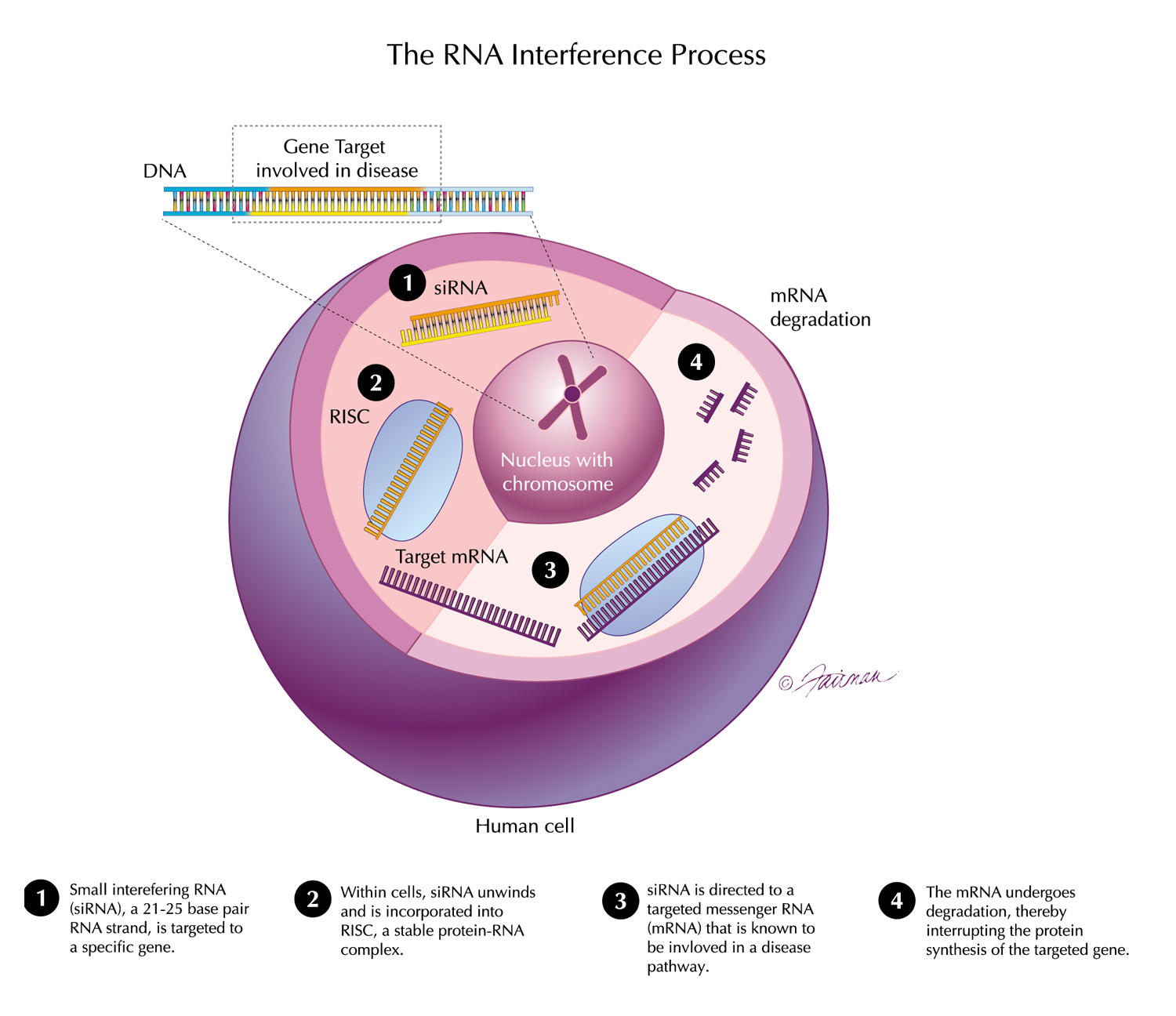
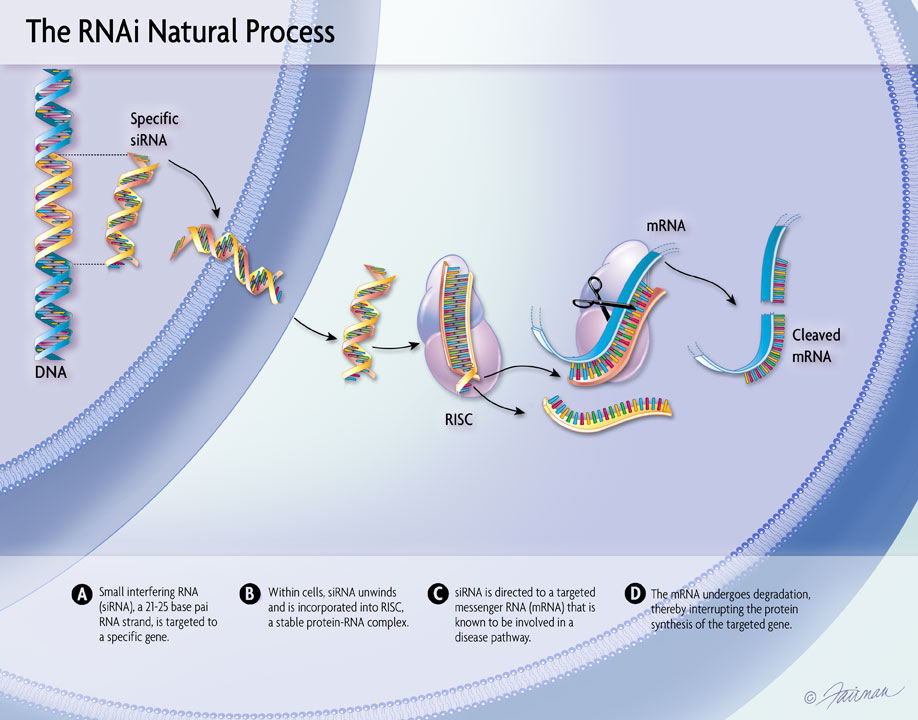
07 Apr, 2022 New Results from the ILLUMINATE-A Study of OXLUMO® (lumasiran) Presented at NKF Spring Meetings
New findings from a 24-month analysis of the ongoing ILLUMINATE-A Phase 3 study of OXLUMO® (lumasiran) in patients with primary hyperoxaluria type 1 (PH1) were presented during the National Kidney Foundation (NKF) Spring Clinical Meetings (April 6-10, 2022).
Additional findings from a post hoc analysis of the impact of baseline urinary oxalate on response to lumasiran in patients with PH1 in the ILLUMINATE-A study were presented.