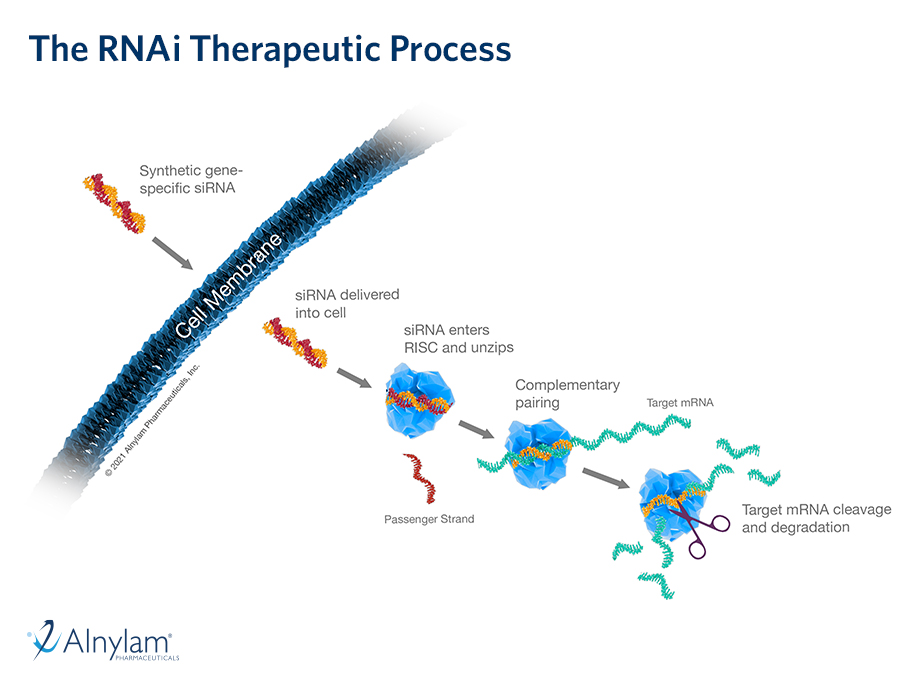
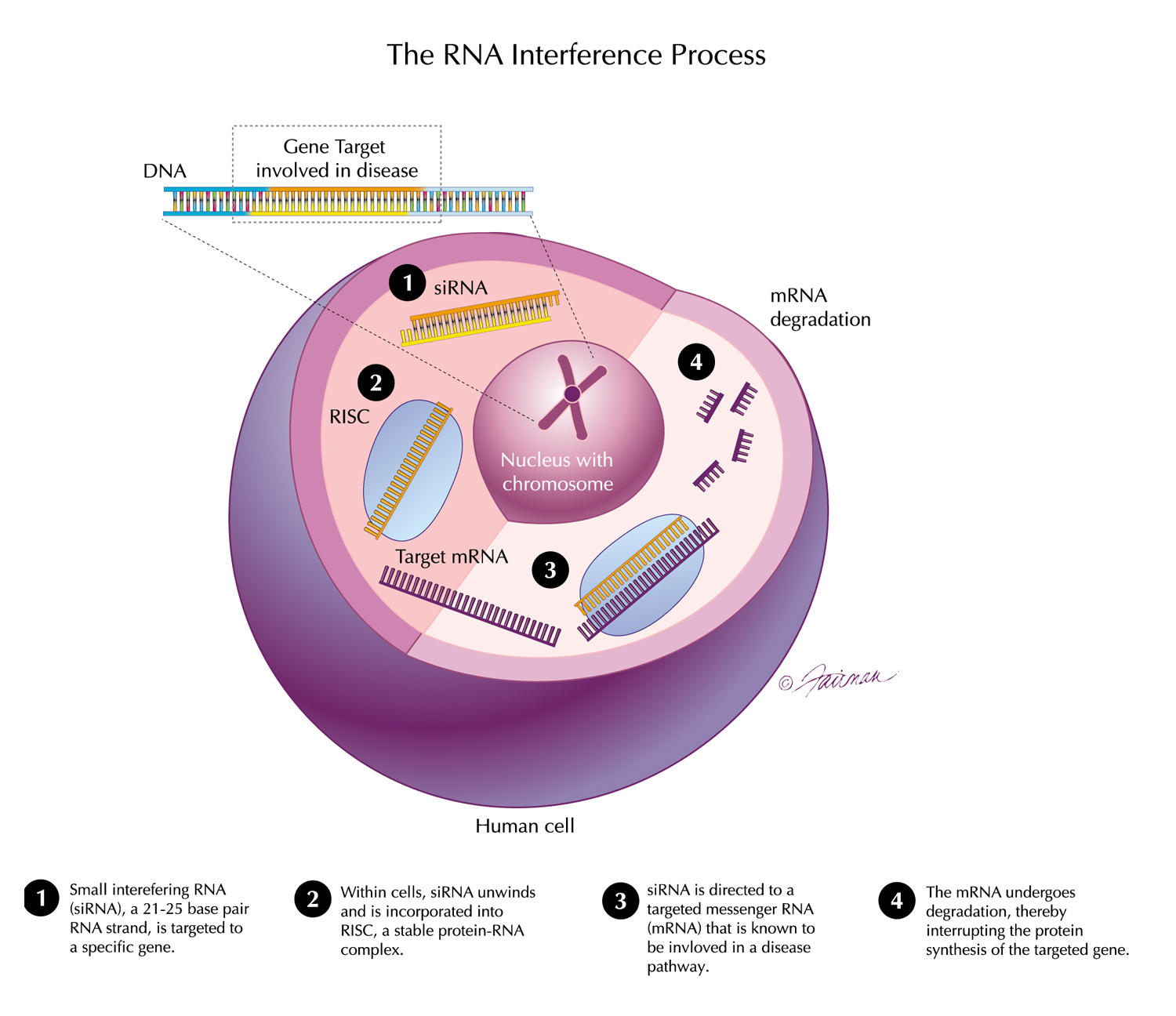
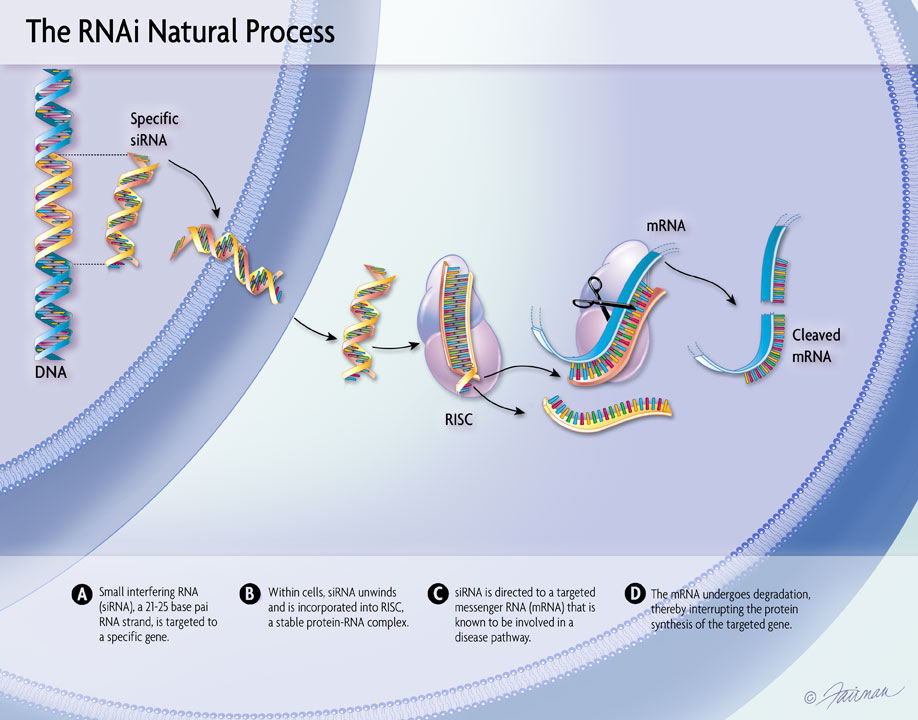
13 Dec, 2021 New Long-Term Results for GIVLAARI® (givosiran) Presented at ASH Annual Meeting & Exposition
Findings from a 36-month analysis of the ENVISION Phase 3 study of GIVLAARI® (givosiran) in patients with acute hepatic porphyria (AHP) were presented during the 63rd American Society of Hematology (ASH) Annual Meeting & Exposition (Dec. 11-14). The data, which encompass the 6-month double-blind and 30-month open label extension (OLE) periods, further confirm that long-term dosing with GIVLAARI provides sustained and continuous benefit to patients with AHP, while maintaining an acceptable and consistent safety profile.
Long-term use of GIVLAARI during the OLE period demonstrated a durable response with efficacy across a wide range of clinical parameters, including sustained reductions in the annualized rate of composite porphyria attacks and hemin use, sustained lowering of ALA and PBG levels and improvements in physical functioning and QOL.